Amiel, D. Ben Abba; Okshtein, H. ; Alshanski, I. ; Hayouka, Z. ; Yitzchaik, S. ; Hurevich, M. .
Expeditious Synthesis Of Multiglycosylated Peptides For Sensing Of Listeria Monocytogenes.
Journal of Medicinal Chemistry 2025,
68, 26513-26524.
Publisher's VersionAbstractGlycosylation is among the most common posttranslational
modifications of proteins. There is a great synthetic and
practical difficulty in the assembly and deprotection of glycopeptides.
State-of-the-art methods for glycopeptide synthesis are wasteful of
glycosylated amino acids, are slow, and suffer from low yields. These
shortcomings hamper accessibility to multiply glycosylated peptides.
We report the accelerated, high-shear stirring-assisted synthesis of
multiply O-glycosylated peptides. The equimolar assembly was
streamlined with deacetylation to provide multiglycosylated peptides
at high purity. Cadherin-derived multiglycosylated peptides synthesized
in large quantities provided selective Listeria monocytogenes
electrochemical biosensing.
We report the accelerated, high-shear stirring-assisted synthesis of
multiply O-glycosylated peptides

Shitrit, A. ; Sukhran, Y. ; Tverdokhleb, N. ; Chen, L. ; Dianat, A. ; Gutierrez, R. ; Körbel, S. ; Croy, A. ; Cuniberti, G. ; Hurevich, M. ; et al. Monosaccharide-Derived Enantioselectivity In Swcnt Chemoresistive Voc Sensing.
Chemistry-A European Journal 2025,
2025, e02553.
Publisher's VersionAbstract
Semiconducting single-walled carbon nanotubes (sc-SWCNTs) are of great potential for vapor sensing. However, sc-SWCNTs lack recognition features for discriminating between sparsely functionalized moieties, molecules with similar structural features, and enantiomer pairs. This becomes a major setback in discriminating between volatile organic compounds (VOCs). Here, we used two galactosides decorated with aromatic groups as a recognition layer in chemoresistive sc-SWCNT sensors to produce chiral preference toward six terpenoid enantiomers. The multichirality and multifunctionality of a monosaccharide scaffold were exploited to maximize the limited interacting features associated with VOCs. The developed system establishes a robust and tunable platform for enantioselective gas sensing.
A chemoresistive sensor based on semiconducting single-walled carbon nanotubes modified with multichiral and multifunctional substituted monosaccharides displays intriguing chiral preference. The Manuscript is highlighted in ChemistryViews by Wiley 10.1002/chemv.202500101

Chen, L. ; Sandonas, L. M. ; Traber, P. ; Dianat, A. ; Tverdokhleb, N. ; Hurevich, M. ; Yitzchaik, S. ; Gutiérrez, R. ; Croy, A. ; Cuniberti, G. .
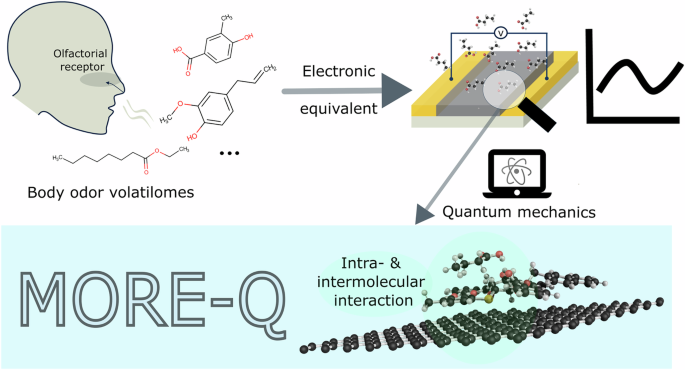
More-Q, A Dataset For Molecular Olfactorial Receptor Engineering By Quantum Mechanics.
Scientific Data 2025,
12, 324.
Publisher's VersionAbstractWe introduce the MORE-Q dataset, a quantum-mechanical (QM) dataset encompassing the structural and electronic data of non-covalent molecular sensors formed by combining 18 mucin-derived olfactorial receptors with 102 body odor volatilome (BOV) molecules. To have a better understanding of their intra- and inter-molecular interactions, we have performed accurate QM calculations in different stages of the sensor design and, accordingly, MORE-Q splits into three subsets: i) MORE-Q-G1: QM data of 18 receptors and 102 BOV molecules, ii) MORE-Q-G2: QM data of 23,838 BOV-receptor configurations, and iii) MORE-Q-G3: QM data of 1,836 BOV-receptor-graphene systems. Each subset involves geometries optimized using GFN2-xTB with D4 dispersion correction and up to 39 physicochemical properties, including global and local properties as well as binding features, all computed at the tightly converged PBE+D3 level of theory. By addressing BOV-receptor-graphene systems from a QM perspective, MORE-Q can serve as a benchmark dataset for state-of-the-art machine learning methods developed to predict binding features. This, in turn, can provide valuable insights for developing the next-generation mucin-derived olfactory receptor sensing devices.
Graphical representation of the motivation for developing MORE-Q dataset (Molecular Olfactorial Receptor Engineering by Quantum mechanics). Bio-electronic noses (top right panel) are designed as an electronic equivalent to the olfactory system (top left panel), e.g. for sensing body odor volatilomes (BOV). The MORE-Q dataset offers a comprehensive collection of quantum-mechanical properties and structural data that accurately describe intra- and intermolecular interactions in molecular sensors, see lower panel.
Okshtein, H. ; Alshanski, I. ; Kikkeri, R. ; Yitzchaik, S. ; Hurevich, M. .
Sialylated Glycan-Based Impedimetric Biosensing For The Detection Of Vibrio Cholerae Biomarkers In Cell Culture Media.
Materials Advances 2025,
6, 8071-8077.
Publisher's VersionAbstract
Cholera is a severe infectious disease caused by Vibrio cholerae. The disease primarily spreads through contaminated food and water sources; it remains a significant global health concern. The pathogenesis of Vibrio cholerae is facilitated by the secreted neuraminidase, Vibrio cholerae neuraminidase (VCNA). This neuraminidase cleaves host cell surface sialic acids, which leads to bacterial colonization and infection progression. This study presents the development of a label-free VCNA biosensor based on electrochemical impedance spectroscopy. The biosensor relies on synthetic sialosides that form self-assembled monolayers on gold electrodes. The system demonstrated selective detection of VCNA activity through distinct impedance variations corresponding to the enzymatic cleavage of the sialoside substrates. The VCNA activity was evaluated under varying environmental conditions, including different media and pH values. This approach provides insights into developing robust biosensing platforms for bacterial detection, offering potential applications in various diagnostic and monitoring systems.
This study presents the development of a label-free VCNA biosensor based on electrochemical impedance spectroscopy. The biosensor relies on synthetic sialosides that form self-assembled monolayers on gold electrodes. The system demonstrated selective detection of VCNA activity through distinct impedance variations corresponding to the enzymatic cleavage of the sialoside substrates.

Bakhatan, Y. ; Sukhran, Y. ; Tsau, L. - S. ; Lin, S. - C. ; Wang, C. - C. ; Hurevich, M. .
Acceptor-Adaptive Automated Glycosylation Optimization For Automated Glycan Assembly.
Chem. Eur. J. 2025,
31, e202501249.
Publisher's VersionAbstractOptimization of glycosylation temperatures is essential for automated solid phase glycan assembly. Constructing a reliable optimization system that mimics key reaction components is crucial for increasing the atom economy and the energy efficiency of the process. Although glycosyl acceptors play a pivotal role in glycosylation, evaluating their effect on glycosylation efficiency is nontrivial. While screening various glycosyl acceptors for optimization is impractical, compromising for simple alkyl acceptors produces results of dubious relevance. We demonstrate that optimization with a modified trans-4-aminocyclohexanol-based acceptor accurately reflects the optimal glycosylation temperature of several glycosyl acceptors, overcoming previous limitations. The contextual use of both n-alkyl and glycan-mimetic acceptors for automated optimization enabled translation of optimized glycosylation temperature to highly efficient solid phase synthesis of disaccharides.
The difficulty of finding the optimal glycosylation conditions for automated solid-phase glycan assembly leads to the use of large temperature ranges and excess reagents. This has negative atom and energy efficiency implications. We demonstrate that mirroring glycosylation temperature from solution to solid phase via an automated acceptor-adaptive optimization strategy empowers an efficient and sustainable automated glycan assembly (AGA) process.

Bressler, S. ; Grunhaus, D. ; Aviram, A. ; Rudiger, S. G. D. ; Hurevich*, M. ; Friedler*, A. .
Specific Phosphorylation Patterns Control The Interplay Between Aggregation And Condensation Of Tau-R4.
Org. Biomol. Chem. 2025,
23, 6912-6923.
Publisher's VersionAbstract
Specific phosphorylation patterns regulate the activity of proteins and play a central role in protein self-assembly. In Tau, such patterns drive the formation of disease-related condensates and aggregates. Understanding their functional impact is essential for studying Tauopathies such as Alzheimer’s Disease. Here we show how specific phosphorylation patterns regulate Tau self-assembly and control the interplay between its aggregation and condensation, using a peptide-based approach that allows systematic analysis of libraries of specific phosphorylation patterns at the domain level and is complementary to the current protein-level methods. We applied our methodology to study the effect of specific phosphorylations on the aggregation and condensation of the R4 domain of Tau that is pivotal for its self-assembly, forming the β-helix motif that is common to various Tau patient fibrils. Using advanced phosphopeptide synthesis methods developed in our labs, we generated a library of multi-phosphorylated peptides derived from Tau R4. We found that phosphorylation at Ser341 promotes aggregation, while Ser352 enhances condensation. Phosphorylation at Ser356 inhibits both processes. The source of these different outcomes is the distinct microenvironments around each phosphorylated site. Our results provide a residue-level resolution of how the decision between Tau condensation and aggregation is being made. This was possible by using our peptide-based approach, which is complementary to protein-level method and enables efficient identification of active phosphorylation patterns. These can later be studied at the protein level.
Using advanced phosphopeptide synthesis methods developed in our labs, we generated a library of multi-phosphorylated peptides derived from Tau R4. Our results provide a residue-level resolution of how the decision between Tau condensation and aggregation is being made. The source of these different outcomes is the distinct microenvironments around each phosphorylated site. The manuscript is highlighted on the journal cover.

Shitrit, A. ; Alshanski, D. Israel; Mor, K. ; Rademann, P. Dr. Jörg; Kikkeri, P. Raghavendr; Hurevich, P. Mattan; Yitzchaik, P. Shlomo.
Metal-Mediated Il-8 Binding To Heparan Sulfate Evaluated By Electrochemical Impedance Spectroscopy.
Chem. Eur. J. 2025,
31, e202501011.
Publisher's VersionAbstract
Heparan sulfate (HS) interactions with interleukin 8 (IL-8) are crucial for immune system response. The structural features of the HS and the environmental entities, such as metal ions, can regulate these interactions. However, it is challenging to evaluate the effect of each parameter on the interactions because of low accessibility to well-defined saccharides and the lack of characteristic features to be determined by analytical tools. We evaluated the effect of the HS structural features on IL-8 binding affinity utilizing electrochemical impedance spectroscopy (EIS) and X-ray photoelectron spectroscopy (XPS). We showed that the metal ions Ca2+ and Mg2+ dissimilarly mediate the interactions of HS and IL-8 in structure-dependent manner of the HS. We showed that in all glycans, a positive synergistic effect on IL-8 binding was observed. For several glycans, the presence of ions resulted in a dramatic increase in the affinity to IL-8, while for other glycans, a milder effect was observed. This demonstrated that both structural motifs and environmental features are crucial for maintaining the interactions between the HS and IL-8.
We evaluated the effect of the HS structural features on IL-8 binding affinity utilizing electrochemical impedance spectroscopy (EIS) and X-ray photoelectron spectroscopy (XPS). We showed that the metal ions Ca2+ and Mg2+ dissimilarly mediate the interactions of HS and IL-8 in structure-dependent manner of the HS.
